Purpose: The purpose of this experiment to observe the diffusion of solutes and the osmosis of water through a semipermeable membrane.
Hypothesis: This lab will show the diffusion of particles through a semi-permeable membrane. Large molecules will not be able to pass through the membrane but water molecules will be able to. I think the mystery solution will resemble the gold colored solution.
Materials:
Three 500 mL beakers
One 1500 mL Graduated Cylinder
150 g Table Sugar
10 g Corn Starch
Dialysis tubing
Distilled water
Food Coloring
Balance scale
Three 500 mL beakers
One 1500 mL Graduated Cylinder
150 g Table Sugar
10 g Corn Starch
Dialysis tubing
Distilled water
Food Coloring
Balance scale
Procedure:
1. Measure out 10 g of starch on balance scale and add to 990 mL of water to create a 1% solution.
2. Measure out 150 g of sugar (sucrose) to 850 mL of water to create a 15% solution.
3. Place both solutions on hot plates add a magnetic stirring rod to stir fully for 8-10 minutes.
4. Cut 12 cm of dialysis tubing, fold and tie at bottom.(x3)
5. Add 15 mL of each solution to dialysis tubing, and fold and tie top.
6. Weigh and observe dialysis tubing.
7. Add water to a beaker until dialysis tubing is completely submerged, for us, 240 mL.
8. Add 12 drops of food coloring to beaker.
9. Cover beaker with plastic bag and tie off top.
10. Observe and weigh tubing after 15 minutes.
11. Observe and weigh tubing after one day.
12. Cut tubing and take pH level.
1. Measure out 10 g of starch on balance scale and add to 990 mL of water to create a 1% solution.
2. Measure out 150 g of sugar (sucrose) to 850 mL of water to create a 15% solution.
3. Place both solutions on hot plates add a magnetic stirring rod to stir fully for 8-10 minutes.
4. Cut 12 cm of dialysis tubing, fold and tie at bottom.(x3)
5. Add 15 mL of each solution to dialysis tubing, and fold and tie top.
6. Weigh and observe dialysis tubing.
7. Add water to a beaker until dialysis tubing is completely submerged, for us, 240 mL.
8. Add 12 drops of food coloring to beaker.
9. Cover beaker with plastic bag and tie off top.
10. Observe and weigh tubing after 15 minutes.
11. Observe and weigh tubing after one day.
12. Cut tubing and take pH level.
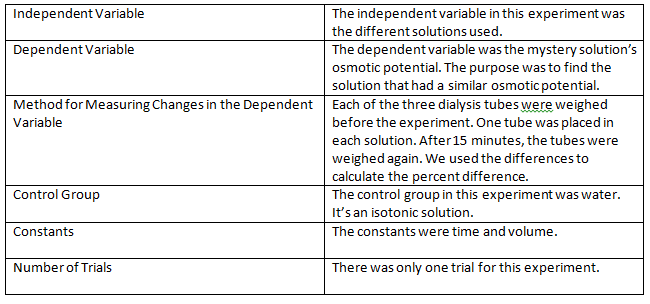
Methods Summary Chart:
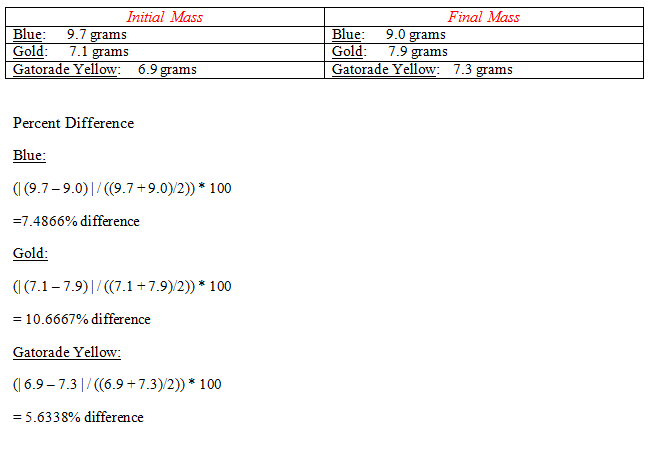
Results:
Discussion/Conclusion:
A. The data show that the mystery solution closely resembled the Gatorade yellow solution. The Gatorade yellow had a 5.6% percent change. This solution moved from a region of high water concentration to one of low water concentration from a hypotonic region (low solute concentration) to a hypertonic region (high solute concentration).
B. My data refutes the hypothesis. I predicted that the mystery solution would closely resemble the gold colored solution. The data shows that the mystery solution was closest to the Gatorade Yellow solution.
C. During this experiment the volumes of the solutions weren't accurately measured out. Although we did our best to accurately measure same amounts of the different solutions we didn't really get the same amounts for each of the solutions. If we were to conduct the experiment again we would use graduated cylinders to get accurate measures for the different solutions.
A. The data show that the mystery solution closely resembled the Gatorade yellow solution. The Gatorade yellow had a 5.6% percent change. This solution moved from a region of high water concentration to one of low water concentration from a hypotonic region (low solute concentration) to a hypertonic region (high solute concentration).
B. My data refutes the hypothesis. I predicted that the mystery solution would closely resemble the gold colored solution. The data shows that the mystery solution was closest to the Gatorade Yellow solution.
C. During this experiment the volumes of the solutions weren't accurately measured out. Although we did our best to accurately measure same amounts of the different solutions we didn't really get the same amounts for each of the solutions. If we were to conduct the experiment again we would use graduated cylinders to get accurate measures for the different solutions.