Purpose: The purpose of this experiment was to determine the effect of the enzyme catalase found in liver on the decomposition of 6% hydrogen peroxide into water and oxygen gas.
Hypothesis: If 6% hydrogen peroxide is added to liver in a room temperature environment the catalase enzyme found in liver will decompose the hydrogen peroxide into water and oxygen gas.
Hypothesis: If 6% hydrogen peroxide is added to liver in a room temperature environment the catalase enzyme found in liver will decompose the hydrogen peroxide into water and oxygen gas.
Materials:
5 test tubes
1 test tube rack
Hydrogen peroxide
Distilled water
Raw beef liver
Boiling water bath (hot plate, water, beaker)
Ice Bath (ice, water, cooler)
5 test tubes
1 test tube rack
Hydrogen peroxide
Distilled water
Raw beef liver
Boiling water bath (hot plate, water, beaker)
Ice Bath (ice, water, cooler)
Procedure:
1. You will prepare the contents as outlined below for each corresponding test tube:
a. Test Tube #1 - Water
b. Test Tube #2 - Water and Liver
c. Test Tube #3 - Water, Liver, and Hydrogen Peroxide
d. Test Tube #4 - Water, Liver(that has been boiled for several minutes), and Hydrogen Peroxide
e. Test tube #5 - Water, Liver(that has been cooled on ice for several minutes), and Hydrogen Peroxide
*Very Important: Do NOT add the Hydrogen Peroxide to Test Tubes #3-5 until you are running the experiment for that test tube.
2. The volume of water of the water will always be 5 mL.
3. The size of the piece of liver should be approximately the same and relatively small.
4. Place test tubes 4 and 5 in ice/ hot water bath as soon as you prepare it.
5. Test ONE test tube at a time.
a. Set up your test tube.
b. Add 5 mL of Hydrogen Peroxide (if indicated)
c. Record you observations ( Approximately 10-15 minutes)
d. Interpret what has happened to the enzyme
1. You will prepare the contents as outlined below for each corresponding test tube:
a. Test Tube #1 - Water
b. Test Tube #2 - Water and Liver
c. Test Tube #3 - Water, Liver, and Hydrogen Peroxide
d. Test Tube #4 - Water, Liver(that has been boiled for several minutes), and Hydrogen Peroxide
e. Test tube #5 - Water, Liver(that has been cooled on ice for several minutes), and Hydrogen Peroxide
*Very Important: Do NOT add the Hydrogen Peroxide to Test Tubes #3-5 until you are running the experiment for that test tube.
2. The volume of water of the water will always be 5 mL.
3. The size of the piece of liver should be approximately the same and relatively small.
4. Place test tubes 4 and 5 in ice/ hot water bath as soon as you prepare it.
5. Test ONE test tube at a time.
a. Set up your test tube.
b. Add 5 mL of Hydrogen Peroxide (if indicated)
c. Record you observations ( Approximately 10-15 minutes)
d. Interpret what has happened to the enzyme
Methods Summary Chart:
Results:
Discussion/Conclusion:
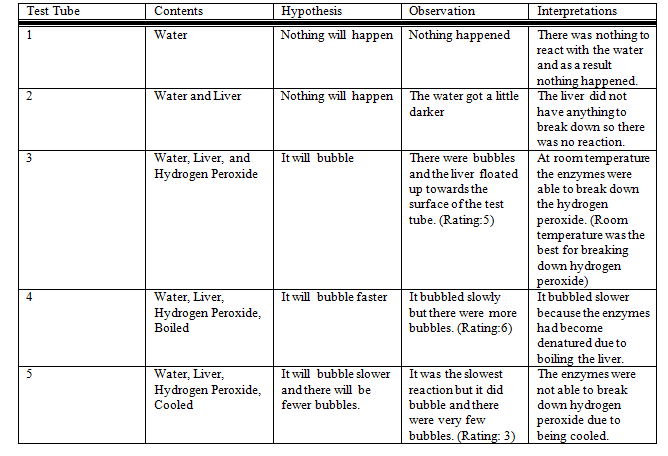
A. The data shows that enzymes work best at room temperature. When the liver was boiled, the catalase enzymes were denatured and therefore unable to break down the hydrogen peroxide. There was also a reduced reaction in the liver enzymes due to the liver being put in an ice bath.
B. The data supports my hypothesis because the enzymes found in liver were able to decompose hydrogen peroxide into hydrogen and oxygen at a reasonably fast rate.
C. A possible source of error could be the scale at which we measured the reaction rate. Every group had a different way of measuring the reaction. We measured the reaction by how many bubbles were produced and the rate at which the bubbles were produced. Another source of error is the how the rate of reaction was measured. It might not be accurate because each step was not repeated to ensure accuracy. Some methods that could eliminate these errors are a scale that we could measure the reaction rate by and having more than one trial to accurately determine the temperature at which the enzymes found in liver work best to decompose hydrogen peroxide.
A. The data shows that enzymes work best at room temperature. When the liver was boiled, the catalase enzymes were denatured and therefore unable to break down the hydrogen peroxide. There was also a reduced reaction in the liver enzymes due to the liver being put in an ice bath.
B. The data supports my hypothesis because the enzymes found in liver were able to decompose hydrogen peroxide into hydrogen and oxygen at a reasonably fast rate.
C. A possible source of error could be the scale at which we measured the reaction rate. Every group had a different way of measuring the reaction. We measured the reaction by how many bubbles were produced and the rate at which the bubbles were produced. Another source of error is the how the rate of reaction was measured. It might not be accurate because each step was not repeated to ensure accuracy. Some methods that could eliminate these errors are a scale that we could measure the reaction rate by and having more than one trial to accurately determine the temperature at which the enzymes found in liver work best to decompose hydrogen peroxide.